Last week I attended parts of the Ohio Craft Brewer’s conference, held at the Hyatt Regency in downtown Columbus. For me the most interesting aspect of the conference was Tom Shellhammer’s keynote address [1]. Shellhammer, the Norwester Professor of Fermentation Science at Oregon State University, is internationally recognized for his research on hop chemistry. His presentation at OCBC focused entirely on the chemistry of dry hopping (adding hops to the beer during conditioning and/or fermentation). As readers of this blog will know, dry hopping is a brewing technique that figures prominently in many styles, most notably American IPAs of every description. Shellhammer’s talk featured three vignettes, each of which had a clear take home message that I attempt to summarize below. I’ve done my best to keep the chemistry at an accessible level.

When more is not always more
The first segment of the talk asked the question, does using larger quantities of dry hops lead to more hop aroma? To answer this question Shellhammer and his colleagues started with base pale ale (4.75% abv) that was very lightly hopped (19 IBU), and dry hopped it with whole cone Cascade hops for 24 hours [2]. The variable they chose to explore was the quantity of dry hops, which ranged from 2 g/L to 16 g/L. Translated into units that would be familiar to metrically challenged American homebrewers, the lowest rate of 2 g/L translates to 1.3 ounces per 5 gallon batch (0.5 lbs/bbl), while the highest rate, 16 g/L, is a whopping 10.7 ounces per 5 gallon batch (4 lbs/bbl). The effects of dry hopping on the finished beer were analyzed using both analytical instrumentation and a trained panel of tasters.
The first conclusion is that above 8 g/L the extraction of hop oils and the aroma contributions from those oils are nearly saturated. To use Shellhammer’s language dry hopping with more than 8 g/L (1.1 oz/gal or 2.1 lbs/bbl) is an inefficient use of raw materials [3]. As a chemistry professor this strikes me as reasonable conclusion. As Tom noted during his talk, the most aromatic components of hop oil have the lowest solubility in water, which limits the amount you can extract. His second finding was more surprising. The oils responsible for citrus aromas saturate earlier, closer to 4 g/L, than the oils responsible for herbal/tea aromas and the compounds that impart bitterness (humulinones and polyphenols). The implications are best summed up in the conclusion of a paper published by Shellhammer and his student Scott LaFontaine [3]:
Adding more hops by static dry-hopping does not simply lead to increased aroma intensity but also changes aroma quality in the finished beer. Dry-hopping rates >8 g/L lead to hop aromas that were more herbal/tea in quality than citrus. To maintain a more balanced hop aroma quality this study suggests using a static dry-hopping rate between 4 and 8 g/L.
How does this compare with dry hopping rates found in practice? I’ve read that Russian River uses approximately 1 lb of dry hops per barrel in Pliny the Elder. While that seemed like a ridiculous amount in the early part of this century, nowadays it doesn’t seem out of the ordinary. Shellhammer’s research would seem to indicate this is still in a range that is useful. Personally, the largest charge of dry hops I’ve used is 4 ounces in a 5 gallon batch, but my buddy Chris Mercerhill once dry hopped with a pound of hops just to see how it would turn out. That experiment, dubbed “The Pounder” also confirmed the notion more is not always better.
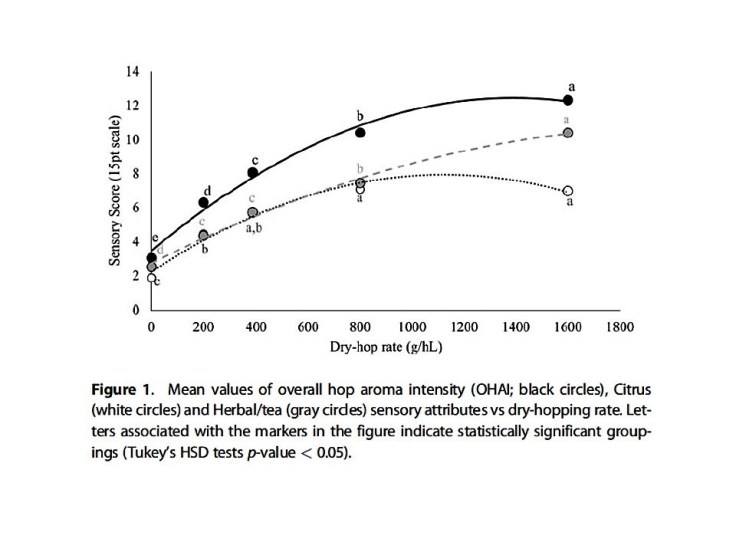
A figure taken from reference [3] showing how the herbal/tea like aromas (gray circles) continue to increase after the citrus aromas (white circles) saturate.
Does dry hopping lead to bitterness?
Until very recently brewers and brewing scientists attributed hop bitterness almost entirely to isomerized alpha acids that are produced in the boiling wort. The alpha acids found in hops are themselves not particularly bitter and have very little solubility in water, but in the harsh conditions of the boil they are isomerized, which in layman’s terms means that the molecule changes its shape (and polarity) by rearranging its atoms. This change increases both bitterness and solubility [4]. At fermentation temperatures where dry hopping occurs there is not enough heat to isomerize the alpha acids, so the conventional thinking was that dry hopping does not add bitterness. For example, the homebrewing calculator that I use assumes no IBU contribution from dry hopping.
With the recent trend toward intensely fruity IPAs with minimal bitterness, brewers have reduced the hops added to the boil (in some cases to almost nothing), while increasing the dry hopping rates significantly. This unprecedented approach to brewing has led brewers and scientists like Shellhammer to rethink the old assumptions. His research shows that compounds called humulinones and polyphenols can make significant contributions to bitterness in heavily dry-hopped beers [5]. Unlike the isomerized alpha acids these compounds do not need to be boiled to become bitter. Furthermore, because the humulinones are formed when (non-isomerized) alpha acids are oxidized, this effect is more dramatic when using old hops that have partially oxidized. What’s the take home message? Don’t put much stock in the low IBU numbers associated with hazy/juicy NEIPAs and if you do want to minimize bitterness seek out brewers that dry hop with the freshest possible hops.
Hop Creep
The final, and arguably most interesting, segment of the talk explored the phenomenon of hop creep. Never heard of hop creep, don’t feel bad, you would have had plenty of company in the audience at the Ohio Craft Brewer’s Conference. It refers to the process whereby additional fermentation is triggered by dry hopping. Shellhammer described an experiment, which is summarized in the graph below taken from his 2018 article in the Journal of Agricultural and Food Chemistry [6].

This plot summarizes Shellhammer’s fascinating experiments with hop creep in Coors Banquet Beer. It was taken from reference [6].
In this study he took four samples of Coors Banquet beer. To one beer he added Cascade hop pellets, to another he added yeast, to a third he added both hops and yeast, while the fourth was not modified. The y-axis of the graph above represents the levels of unfermented sugars in the beer (gravity in brewer’s parlance). The sugar level in the control sample shows almost no change with time, holding steady at 3.5° Plato for 40 days. The beer to which yeast was added shows a modest decrease in sugar levels, perhaps not too surprising, after all Coors doesn’t make the world’s driest beer. The beer to which hops are added shows a slight rise in sugar levels that holds fairly constant for the duration of the experiment. It turns out there are low levels of sugar in hops that can explain this rise. The real shocker is the fourth beer, where both hops and yeast are added. The gravity (sugar levels) of that beer drop by 50% to ~1.7° Plato over the 40-day duration of the experiment!
The important take home point is that additional fermentation only occurs when you add both yeast AND hops. What in God’s green earth could explain this behavior? It turns out there are enzymes in hops that can break down the unfermentable long-chain sugars to simple sugars, much like the enzymes in barley that carry out the same function during the mashing stage. This enzymatic activity doesn’t make much difference if there is no yeast present (sugar is sugar), but in the presence of yeast the newly produced simple sugars are subsequently fermented to ethanol and CO2. Hop creep is only an issue in dry hopping, because if the hops are added to the boiling or near-boiling wort the high temperatures will denature (destroy) the hop enzymes.
The implications of hop creep can be significant, especially for hazy NEIPAs which often contain suspended yeast and depend on high levels of dry hopping to pack in the hop aromas:
- If the beer has already been packaged the additional CO2 produced during fermentation can lead to exploding cans/bottle bombs.
- A boost in abv will be observed, in the Coors Banquet Beer experiment the abv increased by 1.3%
- Additional diacetyl (an unwanted off-flavor that tastes of imitation butter) is produced. Tests in Shellhammer’s lab showed diacetyl levels rose from 25 ppb to 200 ppb due to the bump in fermentation triggered by dry hopping.
The more unfermented sugar in the beer the more problematic hop creep becomes, which means you’re more likely to experience an exploding DIPA can than a Brut IPA.
Think this is just an academic exercise? Try googling the words “exploding beer cans” and see what you find (or read Bryan Roth’s article on Good Beer Hunting). After the conference I was speaking with Mark Richards from Land-Grant Brewing and he told me that recently they had to hold off packaging their most recent hazy IPA (Piña Pants) for an extra week because the gravity unexpectedly kept dropping after the second dry hop addition. They were at a loss to explain the behavior until hearing Shellhammer’s talk, after which everything made sense.
If any brewers out there have stories to share related to dry hopping and this research, feel free to share in the comments section here or on your preferred social media platform. If you are hungry for more details take a look at Tom Shellhammer’s research papers given in the reference section below.
Thanks to Mary MacDonald and Justin Hemminger of the Ohio Craft Brewers Association for allowing me to attend the conference as a member of the media.

References
[1] Not to be confused with the most enjoyable part of the conference, which was sampling barrel aged beers with Angelo Signorino and Dan Eaton at Barley’s Brewcadia on Wednesday evening. The Fate Barleywine was particularly spectacular.
[2] As long-time readers of this site will know, Cascade, the hop that launched the craft beer revolution in America, were developed at Oregon State University. See my earlier post on hops and the American IPA revolution for details.
[3] S. R. Lafontaine, T. H. Shellhammer, “Impact of static dry-hopping rate on the sensory and analytical profiles of beer” Journal of the Institute of Brewing 124, 434−442 (2018).
[4] S. Hieronymous, “For the Love of Hops” Brewers Publications, Boulder (2012).
[5] E. Parkin, T. H. Shellhammer, “Toward understand the bitterness of dry hopped beer” Journal of the American Society of Brewing Chemists 75, 363−368 (2017).
[6] K. R. Kirkpatrick, T. H. Shellhammer, “Evidence of Dextrin Hydrolyzing Enzymes in Cascade Hops (Humulus lupulus)” Journal of Agricultural and Food Chemistry 66, 9121−9126 (2018).
Very interresting. It happened to me, now it is explained! Great!! I added hops and the end of fermentation and the beer turned very sweet!! After a lot of time, it was a great IPA.
Thanks for the information!!!!
Hi! Very interesting! Any info about percentage of essential oils in that Cascade used for the experiment? Thanks!
Yes the essential oil content in the hops was measured. It’s reported in a couple of ways. The total oil content is reported as 2.0 (mL/100 g). Individual oil components are also reported as 1.095 g oils per 100 g of hop matter. The largest component is myrcene at 729 mg, followed by humulene (185 mg) and caryophyllene (95 mg). Interestingly in the dry hopped beers the oils that are present change considerably. Linalool is the largest component followed by myrcene and gerionol.
I’ve never had an exploding bottle in all the years I’ve been home brewing but this hop creep is something to think about. I love the NEIPA style and I do some postal swaps with other Aussie home brewers. In PET bottles I had one not make its destination posted last summer. Reported as damaged. It was a NEIPA bottled from the keg. I’m wondering in the 10 days or more in hot weather in transit to Queensland this may have (Hop Creep fermented) to a point that split the bottle maybe. I cant ever know but this is a real possibility after reading this. My NEIPA is pressure fermented self carbonated then kegged and drunk. So I will do some experiments on bottling from the keg then let sit for some time unrefridgerated, especially in summer and see what happens.
As you say it’s hard to impossible to know for sure, but hop creep could well be a contributing factor.
Thanks for sharing this, Pat, and for bringing the science of it within reach of home brewers. I’ve been using the Tilt Hydrometer to measure gravity in my beers for a while, to help me decide when the yeast has really “finished” with the beer. I had noticed (before reading this) that with the beers I have dry-hopped, there was a sudden drop-off of gravity after sometimes days of no change. I suspected something about the hops was triggering that change, and thanks to you I now know what that is!
I’m glad you found this story helpful Michael. Thanks for sharing your own experiences with hop creep.
Hi Pat, Did Thomas make any extrapolations for higher alpha oil hops saturating with smaller amounts due to the higher percentage of oils in his presentation? From a layman’s perspective, it would stand to reason if cascade is 6% alpha acid and saturates around 5 oz per 5 gallon batch, a dose of 12% alpha acid hops would saturate at 2.5 oz?
Also, did he make any mention of if the dry hop saturation rate was dependent or independent of hops used in the boil which should be isomerized?
Thanks for sharing this info! I have definitely experienced hop creep but just thought Beersmith calculations were probably just way off.
After reading this, I can suddenly explain why my bottle conditioned NEIPA had such a high carbonation. I went back and forth through my calculations but couldn’t find a mistake. I told myself that I must have done something wrong and added too much sugar. So hop creep surely happened to me. I guess I‘ll wait a little longer before botteling or rather use kegs instead (so I can release some of the preassure if too much builds up). Thabks for the article.
I’m glad you found it useful. The two approaches you outline are the only avenues I can think of to avoid (or at least mitigate) hop creep.
thanks for sharing. this was helpful, unlike most shit on the internet